We were the first to set up specific laboratory assays for DOACs in our country, for dabigatran in 2011, for rivaroxaban in 2012 and for apixaban in 2015. We also, to increase the national awareness, authored an article in the Finnish Medical Society Journal to encourage rational introduction of these new drugs ( 2), and national web-based guidelines have been gathered (accessible at in Finnish). Nevertheless, questions regarding anticoagulation of these vulnerable patients are often encountered, and we could not recommend DOACs for these patients at this early stage. 
In the phase III trials of the DOACs, relevant patient groups, such as patients with kidney or liver failure, patients having antiplatelet therapy, patients encountering bleeds and patients with active malignancy were excluded ( 1). When the direct oral anticoagulants (DOACs) became available from 2008 onwards, initially, we were not confident on the overall safety of these new drugs, in the absence of the monitoring of the drug effect. The Unit collaborates with the laboratory at multiple levels on daily basis. 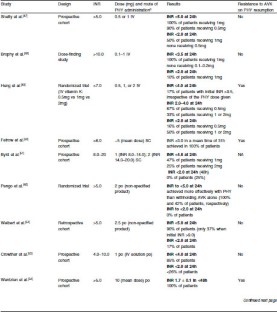
Three to four doctors and 3–4 nurses work at the Unit. The European Association of Haemophilia and Allied Disorders EUHANET project assigned the European Haemophilia Comprehensive Care Centre, EHCCC status to the Unit in 2014. ‘All’ consultations refer to the ones received to our unit, while ‘handled’ consultations refer to the online performed consultations, most urgent being the phone call consultations. The majority of the increased number of consultations are due to problems related to anticoagulation. Interaction, collaboration and continuous education between clinic and laboratory is increasingly needed, under the era of rapid change of traditions in anticoagulation management.įigure 1 The increasing (55%/5 years) number of consultations to the clinical Coagulation Disorders Unit, Department of Hematology and Comprehensive Cancer Centre, Helsinki University Hospital. 
Specific assays for all DOACs are available 24/7 in the core laboratory (Southern Finland). Finland benefits from centralized laboratory networks, offering harmonized services throughout our rather sparsely populated country. Laboratory monitoring of warfarin and heparins are well established, while under specific emergency conditions DOACs require specific assays, the availability of which may be limited. While DOACs are increasingly used in common indications for anticoagulation, including non-valvular atrial fibrillation (AF), deep vein thrombosis (DVT) and pulmonary embolism (PE), traditional anticoagulants, either warfarin or heparins, remain the primary choice for patients with cardiac valvular replacement, severe thrombophilia, pediatric, pregnant and many cancer patients. Policy of Dealing with Allegations of Research MisconductĪbstract: Anticoagulation therapy has been undergoing a transition during the last decade with the progressive availability of direct oral anticoagulants (DOACs).Policy of Screening for Plagiarism Process.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
